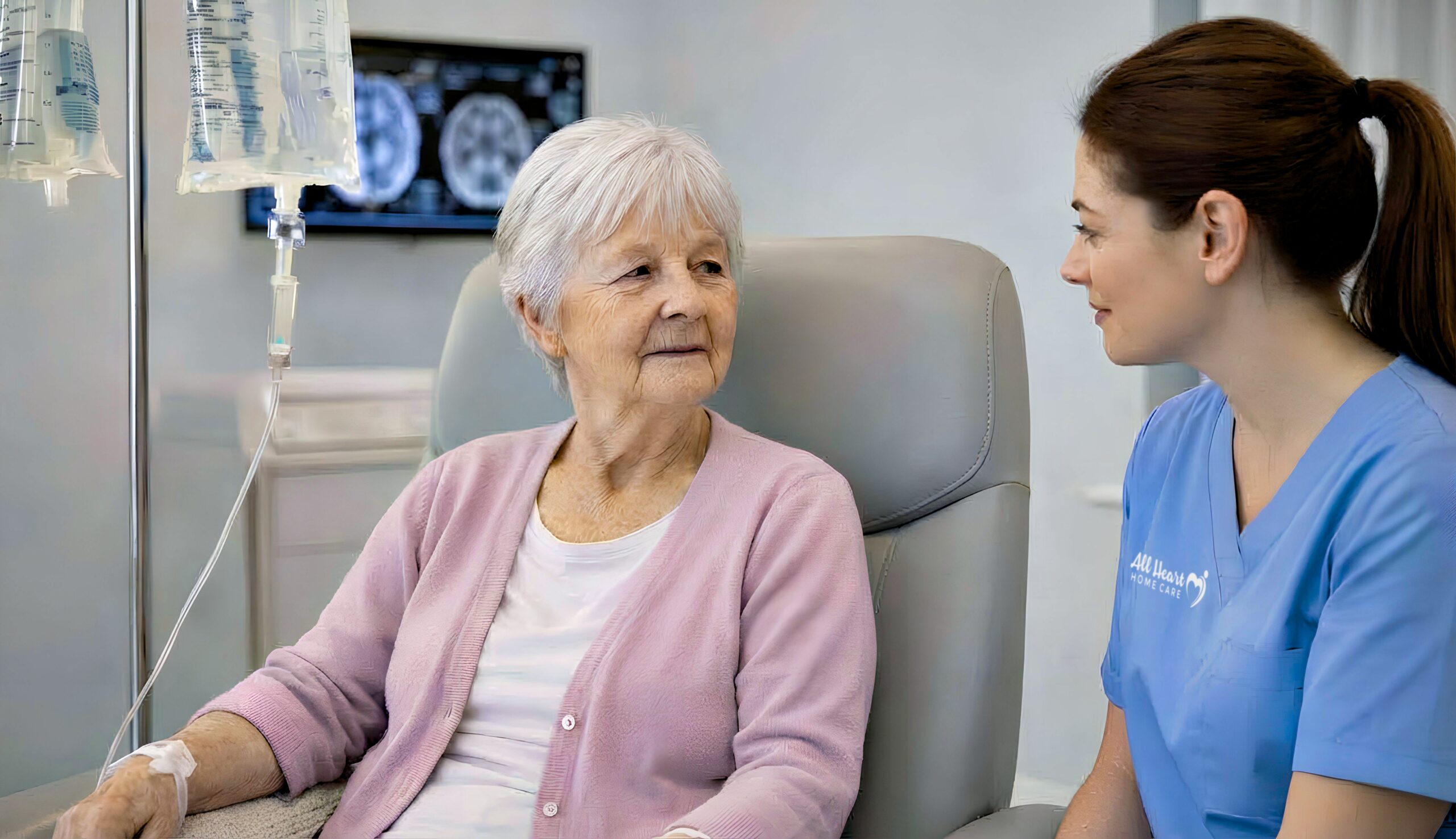
New Alzheimer’s treatments are changing everything for families facing this diagnosis. For the first time in history, we have medications that don’t just treat symptoms — they actually slow the disease itself.
If your loved one was diagnosed with Alzheimer’s five years ago, the only options were medications that temporarily improved symptoms while the disease continued its relentless progression. Today, these new Alzheimer’s treatments give families genuine choices that can preserve months — potentially years — of meaningful time together.
But these new treatments aren’t for everyone. They require early diagnosis, regular infusions, frequent MRI monitoring, and careful observation for side effects. Understanding what’s available — and what it actually means for your family — is essential.
This guide explains the breakthrough Alzheimer’s treatments approved in 2023-2024, who qualifies, what treatment involves, and how home caregivers support families navigating this new era of Alzheimer’s care.
Why These New Alzheimer’s Treatments Are Different
Previous Alzheimer’s medications — donepezil (Aricept), rivastigmine (Exelon), and memantine (Namenda) — work by boosting brain chemicals that support memory and thinking. They can temporarily improve symptoms, but they don’t change the underlying disease. The brain continues to deteriorate.
The new Alzheimer’s treatments work completely differently. They’re monoclonal antibodies that target and remove beta-amyloid plaques — the sticky protein clumps that accumulate in the brains of people with Alzheimer’s and are believed to drive the disease.
By clearing these plaques, the medications slow the rate of cognitive decline. In clinical trials, patients receiving treatment declined more slowly than those receiving placebo, preserving an estimated 4.5 to 7.5 additional months of function over 18 months of treatment.
That may not sound dramatic, but for families, those months represent holidays celebrated together, conversations remembered, and independence maintained. It’s time that matters.
The Two FDA-Approved Disease-Modifying Treatments
Leqembi (Lecanemab)
FDA Approval: July 2023 (traditional approval)
How it works: Leqembi targets and removes beta-amyloid protofibrils — the toxic, soluble form of amyloid believed to be most harmful to brain cells — as well as larger amyloid plaques.
Administration: Intravenous (IV) infusion every two weeks, lasting approximately one hour per infusion. After completing the initial 18-month regimen, patients now have maintenance options: an IV infusion once every 4 weeks OR a weekly subcutaneous (under-the-skin) injection that can be administered at home.
Clinical results: In the CLARITY AD trial involving 1,795 patients, Leqembi slowed cognitive decline by 27% compared to placebo over 18 months. Patients showed statistically significant benefits on measures of cognition, daily function, and quality of life.
Cost: Approximately $26,500 per year. Medicare covers Leqembi for eligible patients enrolled in an approved registry.
Kisunla (Donanemab)
FDA Approval: July 2024 (traditional approval)
How it works: Kisunla targets a modified form of beta-amyloid (pyroglutamate Aβ) found specifically in established amyloid plaques. It’s designed to clear existing plaque deposits from the brain.
Administration: Intravenous (IV) infusion once every four weeks (monthly), lasting approximately 30 minutes. A key advantage: treatment can potentially be stopped once amyloid plaques are cleared to minimal levels, as confirmed by a PET scan — typically after 12-18 months.
Clinical results: In the TRAILBLAZER-ALZ 2 trial involving 1,736 patients, Kisunla slowed cognitive and functional decline by approximately 29% compared to placebo over 76 weeks. This corresponds to a delay in symptom progression of 4.5 to 7.5 months.
Cost: Approximately $32,000 per year for a person of average weight. Medicare coverage is available for patients enrolled in an approved registry.
Leqembi vs. Kisunla: Key Differences
| Leqembi | Kisunla | |
|---|---|---|
| Infusion frequency | Every 2 weeks | Every 4 weeks (monthly) |
| Treatment duration | Ongoing (with maintenance dosing) | Can stop when plaques are cleared (typically 12-18 months) |
| At-home option | Yes (subcutaneous maintenance) | No (IV only) |
| Annual cost | ~$26,500 | ~$32,000 |
| Slowing of decline | ~27% | ~29% |
Both medications have similar efficacy. The choice between them often depends on practical factors: infusion frequency preference, whether the possibility of completing treatment appeals to the patient, and insurance coverage.
Who Qualifies for Treatment
These medications are specifically approved for early-stage Alzheimer’s disease. Not everyone with memory problems is a candidate. If you’re exploring Alzheimer’s and dementia home care options, understanding treatment eligibility is an important first step.
Eligible patients for new Alzheimer’s treatments must have:
- Mild cognitive impairment (MCI) due to Alzheimer’s — Memory and thinking problems that are noticeable but don’t significantly interfere with daily life
- OR mild dementia due to Alzheimer’s — Early-stage dementia, where the person still has significant independence
- Confirmed amyloid pathology — Proof that amyloid plaques are present in the brain, demonstrated through amyloid PET scan or cerebrospinal fluid (CSF) testing. Blood tests (plasma pTau217) are increasingly being used for initial screening.
Who does NOT qualify:
- People with moderate or advanced Alzheimer’s disease
- People with other types of dementia (vascular dementia, Lewy body dementia, frontotemporal dementia)
- People without confirmed amyloid pathology
- People taking blood thinners (anticoagulants) — increased risk of brain bleeding
- People with certain genetic profiles may face restrictions (see below)
The ApoE4 Gene Factor
Before starting treatment, patients undergo genetic testing for the ApoE4 gene — a genetic variant that increases both Alzheimer’s risk and the risk of treatment side effects.
- No ApoE4 copies: Lower risk of side effects — typically good candidates for treatment
- One ApoE4 copy (heterozygous): Moderate risk — treatment generally appropriate with careful monitoring
- Two ApoE4 copies (homozygous): Higher risk of serious side effects — the European Medicines Agency restricts Leqembi use in this group; careful risk-benefit discussion required
Understanding ARIA: The Most Important Side Effect
The primary safety concern with both new Alzheimer’s treatments is ARIA — Amyloid-Related Imaging Abnormalities. This refers to brain swelling (ARIA-E) or small areas of bleeding (ARIA-H) that can occur as amyloid is cleared from the brain.
Key facts about ARIA:
- ARIA occurs in approximately 20-35% of patients
- Most cases are asymptomatic — detected only on routine MRI monitoring
- When symptoms occur, they may include headache, confusion, dizziness, vision changes, nausea, or difficulty walking
- Most ARIA resolves on their own within weeks to months
- Serious or life-threatening ARIA is rare but possible — the FDA has identified fatal cases, prompting enhanced monitoring requirements
MRI monitoring schedule:
Patients receiving these treatments require multiple brain MRIs to monitor for ARIA:
- Baseline MRI before starting treatment
- MRIs before specific infusions (schedule varies by medication)
- Additional MRIs if symptoms develop
- For Leqembi: MRIs recommended before the 3rd, 5th, 7th, and 14th infusions
What Treatment Actually Looks Like
Understanding the practical realities of these new Alzheimer’s treatments helps families make informed decisions.
Before Treatment Begins
- Comprehensive cognitive evaluation to confirm diagnosis and stage
- Amyloid confirmation (PET scan or CSF testing)
- Genetic testing for ApoE4 status
- Baseline brain MRI
- Review of all medications (blood thinners must be stopped)
During Treatment
- Regular infusions: Every 2 weeks (Leqembi) or monthly (Kisunla) at an infusion center or hospital
- Infusion time: Approximately 30-60 minutes plus observation
- MRI monitoring: Multiple scans throughout treatment (schedule varies)
- Regular follow-up: Ongoing cognitive assessments and safety monitoring
- Side effect watch: Families learn to recognize potential ARIA symptoms
Infusion-Related Reactions
The most common side effect (affecting about 25% of patients) is infusion-related reactions during or shortly after the infusion. These typically include:
- Flushing, chills, or fever
- Headache
- Nausea
- Body aches
- Rash
Most reactions are mild to moderate and occur primarily with the first few infusions.
What’s Coming Next: The Future of Alzheimer’s Treatment
Research on new Alzheimer’s treatments continues rapidly. As of 2025, 138 drugs are being studied in 182 clinical trials for Alzheimer’s disease. Some notable developments:
Tau-targeting therapies: While current treatments target amyloid, the next generation may target tau protein — another hallmark of Alzheimer’s. Biogen’s BIIB080 and other tau-targeting drugs are in Phase 2 trials, with major results expected in 2026.
Easier delivery: Subcutaneous (injection) and potentially oral formulations are being developed to reduce the burden of frequent visits to infusion centers.
Blood-based diagnostics: Simple blood tests (like pTau217) are increasingly accurate at detecting Alzheimer’s pathology — matching PET scan results more than 90% of the time. This will make screening more accessible and affordable.
Prevention trials: Studies are underway testing whether these treatments can prevent Alzheimer’s in people who have amyloid buildup but no symptoms yet.
Combination approaches: Future treatment may involve multiple drugs targeting different aspects of the disease, similar to how cancer and HIV are now treated.
How Home Caregivers Support Treatment Success
These new Alzheimer’s treatments don’t eliminate the need for supportive care — they increase it. Treatment success depends on consistent monitoring, transportation to frequent appointments, and vigilant observation for side effects. Whether you need hourly home care for appointment days or 24-hour home care for comprehensive support, professional caregivers play a vital role.
At All Heart Home Care, our caregivers play a vital role in supporting families navigating Alzheimer’s treatment:
✓ Transportation to infusion appointments — Reliable transportation every 2-4 weeks to infusion centers, plus additional trips for MRIs and follow-up visits
✓ Side effect monitoring — Trained to recognize potential ARIA symptoms (headache, confusion, vision changes, dizziness) and alert family members and healthcare providers immediately
✓ Post-infusion observation — Providing companionship and monitoring during the hours following each infusion
✓ Medication management — Ensuring all prescribed medications are taken correctly and tracking any that must be stopped during treatment
✓ Cognitive engagement — Supporting brain-healthy activities that complement medical treatment: puzzles, conversation, reminiscence activities, physical exercise
✓ Daily living support — Helping with meal preparation, personal care, and household tasks so the person with Alzheimer’s can focus on treatment and maintaining quality of life
✓ Communication with healthcare team — Documenting observations, tracking symptoms, and providing detailed reports to physicians
✓ Caregiver respite — Giving family members time to rest while ensuring their loved one receives consistent, attentive care
These medications slow decline — they don’t stop it. Supportive care remains essential for quality of life, safety, and independence throughout the treatment journey.
Important Considerations About New Alzheimer’s Treatments
These new Alzheimer’s treatments are not cures. They slow the progression of Alzheimer’s disease but do not stop or reverse it. Cognitive decline continues, just at a slower rate.
Earlier treatment is better. These medications work best in the earliest stages of disease. If you notice memory changes in a loved one, seek evaluation promptly. Early diagnosis now opens doors that didn’t exist before.
Not everyone benefits equally. Some patients show more improvement than others. Clinical trials measure averages — individual responses vary.
Treatment is a commitment. Regular infusions, frequent MRIs, and ongoing monitoring — treatment requires significant time and coordination.
Access varies. Availability depends on proximity to qualified infusion centers, insurance coverage, and healthcare system capacity. Rural areas may have limited access.
Costs add up. Beyond drug costs, families should anticipate expenses for PET scans, MRIs, genetic testing, infusion center visits, and transportation.
Questions to Ask the Doctor
If you’re considering these new Alzheimer’s treatments for a loved one, here are essential questions for their healthcare provider:
- Is my loved one in the early stage where treatment is likely to help?
- What testing is needed to confirm eligibility?
- What is their ApoE4 status, and how does that affect the risk-benefit calculation?
- Which treatment do you recommend, and why?
- Where will infusions take place, and how do we arrange transportation?
- What’s the MRI monitoring schedule?
- What symptoms should we watch for at home?
- What are the total estimated costs, and what will insurance cover?
- Can current symptom-relief medications continue during treatment?
We’re Here to Help
At All Heart Home Care, we’ve spent 12 years helping San Diego families navigate Alzheimer’s and dementia care. As these new Alzheimer’s treatments become available, we’re here to support families through every stage of the journey — from diagnosis through treatment and beyond.
Whether your loved one is starting one of these new Alzheimer’s treatments or managing the disease with traditional approaches, our caregivers provide the consistent, compassionate support that makes the difference between surviving and thriving.
Call us at (619) 736-4677 for a free in-home consultation.
We’ll assess your loved one’s care needs and create a personalized plan that supports their health, safety, and quality of life — today and as their needs evolve.
Because medical breakthroughs matter most when families have the support to make the most of them.
Resources
- Alzheimer’s Association 24/7 Helpline: 1-800-272-3900
- Alzheimer’s Association Treatment Information: alz.org/treatments
- Leqembi Copay Assistance Program: 1-833-453-7362
- Medicare Coverage Information: medicare.gov
- Clinical Trial Finder: clinicaltrials.gov
References
- U.S. Food and Drug Administration. FDA Converts Novel Alzheimer’s Disease Treatment to Traditional Approval. July 2023. FDA.gov
- U.S. Food and Drug Administration. FDA Approves Treatment for Adults with Alzheimer’s Disease. July 2024. FDA.gov
- van Dyck CH, et al. Lecanemab in Early Alzheimer’s Disease. New England Journal of Medicine. 2023;388:9-21.
- Sims JR, et al. Donanemab in Early Symptomatic Alzheimer’s Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. JAMA. 2023;330(6):512-527.
- Cummings J, et al. Alzheimer’s Disease Drug Development Pipeline: 2025. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. June 2025.
- Alzheimer’s Association. Lecanemab and Donanemab Treatment Information. 2025. alz.org
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Treatment decisions should be made in consultation with qualified healthcare providers who can evaluate individual circumstances. Eligibility for Alzheimer’s treatment, risks, and benefits vary by patient. Always consult your physician before starting or changing any treatment regimen.